The Organization
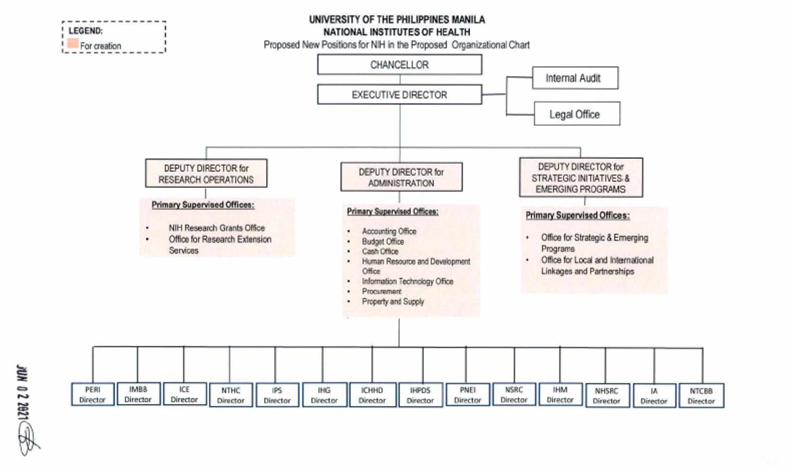
The National Institutes of Health is led by the Executive Director, responsible for the overall supervision and coordination of existing and future research-related units and offices within the NIH. Assisting the Executive Director in these duties are the Deputy Directors and Directors who serve to strengthen the upper management and development of the NIH.
Below is the NIH organizational chart approved by the UP Board of Regents in its 1361st Meeting in June 2021.
News Image: